Apical HCM: a less familial variant that requires a higher index of suspicion
Compared with classical HCM, apical HCM is a more sporadic disease with fewer patients reporting a positive family history. Gene mutations are identified in as few as 13% of cases, compared with 40% in the “classical” form of HCM that causes asymmetrical septal hypertrophy.
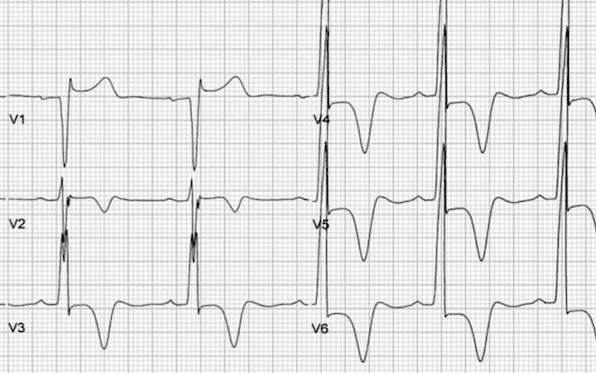
“Giant” negative T waves (> 10mm) are characteristic but not mandatory for diagnosis, with only half of patients presenting with this typical ECG feature.
The absence of “dagger” Q waves means that the ECG of apical HCM can easily be mistaken for uncomplicated LVH. In addition, most cases of apical HCM do not manifest LV outflow obstruction, resulting in fewer presentations of exertional pre-syncope or syncope. Typical diagnostic criteria for HCM are therefore less applicable to the apical variant, and a higher index of suspicion is required for diagnosis.
There are both “pure” (isolated apical) and “mixed” (apical and septal) forms of disease — our patient above has evidence of anteroseptal hypertrophy on echo indicating a mixed form. Note how the LV wall fails to taper off at the apex as it usually does, instead persisting in size or even thickening. Some describe the apical cavity obliteration seen in the pure form as reflecting the shape of an “ace of spades” (hover over image and drag slider to reveal)
Note that imaging of the apex with echo can be challenging. Cardiac MRI is a more sensitive diagnostic tool for detecting subtle apical abnormalities suggestive of early disease.
Inducible ischaemia in apical HCM
As seen in our patient above, severe LV apical thickening can lead to complete cavity obliteration in systole. Such obliteration can result in dynamic small vessel-obstruction in apical segments, causing recurrent ischaemic chest pain with an associated troponin rise. Prolonged, ineffective apical contraction (often into mid-diastole) further exacerbates this ischaemia.
What about our first ECG?
LV voltage criteria are identical between our two ECGs, with the only difference being the depth of T wave inversion. There are two possible explanations for this. One is that in our first ECG, a relative tachycardia results in less pronounced repolarisation abnormalities. The other is that with inducible ischaemia, there is a small degree of “pseudonormalisation” of T wave inversion. Either way, we know that diagnosing OMI in the context of LVH is very difficult and relies mainly on serial ECGs and clinical suspicion. There are no specific criteria for diagnosing OMI in the presence of LVH as there are for LBBB.
- Sporadic genetic mutations, lack of “dagger” Q waves, and an absence of LVOT obstruction means a high index of suspicion is required to diagnose apical HCM
- We must be cautious of diagnosing LVH in the absence of inciting stimuli. HCM is not necessarily a “young person” disease — the mean age of diagnosis is 41
- Inducible ischaemia is common in apical HCM, leading to recurrent exertional chest pain that requires careful symptomatic management